Nevro receives CE mark for full-body MRI conditional labelling with the Senza system - NeuroNews International

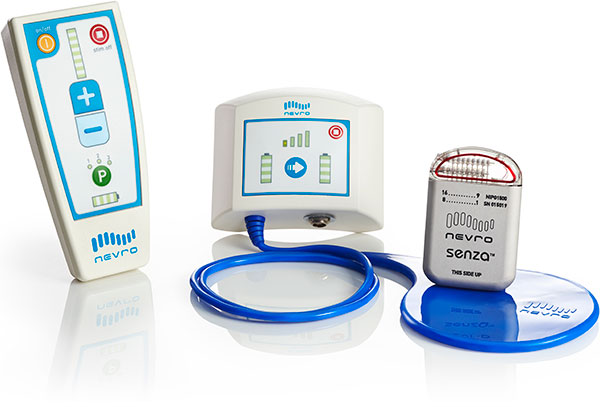
Nevro's Senza Rechargeable Spinal Cord Stimulator for Back and Leg Pain – The World of Implantable Devices
1.5 Tesla and 3 Tesla Magnetic Resonance Imaging (MRI) Guidelines for the Senza II™ Implantable Pulse Generator (IPG2000)
1.5 Tesla and 3 Tesla Magnetic Resonance Imaging (MRI) Guidelines for the SENZA® Neuromodulation Systems

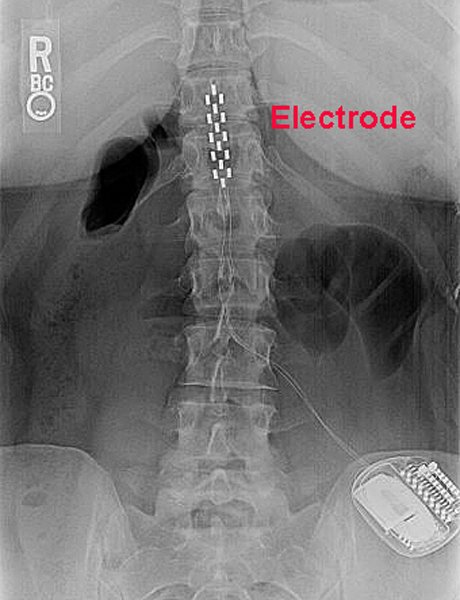
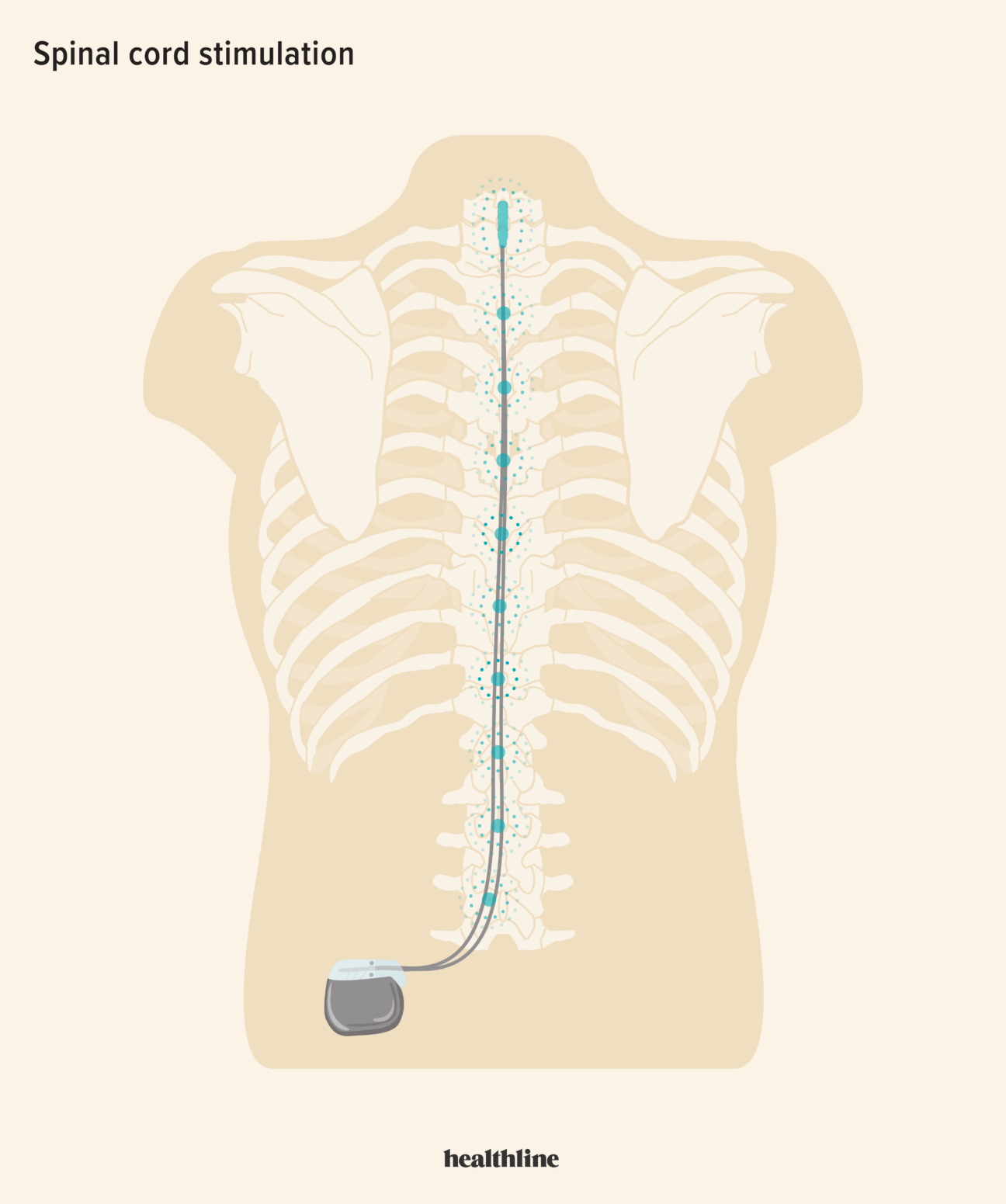
Spinal cord stimulation and rehabilitation in an individual with chronic complete L1 paraplegia due to a conus medullaris injury: motor and functional outcomes at 18 months | Spinal Cord Series and Cases

Nevro Announces FDA Approval of its 10 kHz High Frequency Spinal Cord Stimulation Therapy for Treatment of Chronic Pain Associated with Painful Diabetic Neuropathy (PDN)